 |
 |
 |
| Главная Журналы Популярное Audi - почему их так назвали? Как появилась марка Bmw? Откуда появился Lexus? Достижения и устремления Mercedes-Benz Первые модели Chevrolet Электромобиль Nissan Leaf |
Главная » Журналы » Absorbing materialorganic polymer 1 ... 3 4 5 6 7 8 9 ... 55
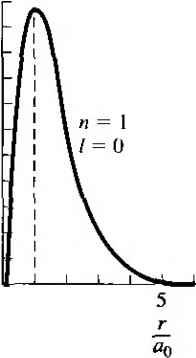  OA - Figure 2.101 The radial probability density function for the one-electron atom in the (a) lowest energy state and (b) next-higher energy state. (From Eisherg and Re snick [4j.} density function for the lowest energy state is plotted in Figure 2.10a. The most probable distance from the nucleus is at r = о, which is the same as the Bohr theory. Considering this spherically symmetric probability function, we may now begin to conceive the concept of an electron cloud, or energy shell, surrounding the nucleus rather than a discrete particle orbiting around the nucleus. The radial probability density function for the next higher, spherically symmetric wave function, corresponding to n -2,1 =0, and m - 0, is shown in Figure 2.10b. This figure shows the idea of the next-higher energy shell of the electron. The second energy shell is at a greater radius from the nucleus than the first energy shell. As indicated in the figure, though, there is still a small probability that the electron will exist at the smaller radius. For the case of n - 2 and / = 1, there are three possible states corresponding to the three allowed values of the quantum number m. These wave functions are no longer spherically symmetric. Although we have not gone into a great deal of mathematical detail for the one-electron atom, three results are important for the further analysis of semiconductor materials. The first is the solution of Schrodingers wave equation, which again yields electron probability functions, as it did for the simpler potential functions. In developing the physics of semiconductor materials in later chapters, we will also be considering electron probability functions. The second result is the quantization of allowed energy levels for the bound electron. The third is the concept of quantum numbers and quantum states, which evolved from the separation-of-variables technique. We will consider this concept again in the next section and in later chapters when we deal with the semiconductor material physics. 2.4.2 The Periodic Table The initial portion of the periodic table of elements may be determined by using the results of the one-electron atom plus two additional concepts. The first concept needed is that of electron spin. The electron has an intrinsic angular momentum, or spin, which is quantized and may take on one of two possible values. The spin is designated by a quantum number 5, which has a value of + or - - . We now have four basic quantum numbers: /?, /, m, and s. The second concept needed is the Pauli exclusion principle. The Pauli exclusion principle states that, in any given system (an atom, molecule, or crystal), no two electrons may occupy the same quantum state. In an aiotn, the exclusion principle means that no two electrons may have the same set of quantum numbers. We will see that the exclusion principle is also an important factor in determining the distribution of electrons among available energy states in a crystal. Table 2.1 shows the first few elements of the periodic table. For the first element, hydrogen, we have one electron in the lowest energy state corresponding to n - I. From Equation (2.71) both quantum numbers / and m must be zero. However, the electron can take on either spin factor + or - . For helium, two electrons may exist in the lowest energy state. For this case, / m = 0, so now both electron spin states are occupied and the lowest energy shell is full. The chemical activity of an element is determined primarily by the valence, or outermost, electrons. Since the valence energy shell of helium is full, helium does not react with other elements and is an inert element. The third element, lithium, has three electrons. The third electron must go into the second energy shell corresponding ion 2. When /2 - 2, the quantum nuinber / may be 0 or I, and when / - 1, the quantum number m may be 1, 0, or 1. In each case, the electron spin factor may be-{- or-. For я = 2, then, there are eight possible quantum states. Neon has ten electrons. Two electrons are in the n - 1 energy shell and eight electrons are in the - 2 energy shell. The second energy shell is now full, which means that neon is also an inert element. From the solution of Schrodingers wave equation for the one electron atom, plus the concepts of electron spin and the Pauli exclusion principle, we can begin to build up the periodic table of elements. As the atomic nuinbers of the elements increase, electrons will begin to interact with each other, so that the buildup of the periodic table will deviate somewhat from the simple method. Table 2.11 Initial portion of the periodic table
2.5 I SUMMARY We considered some of the basic concepts of quantum mechanics, which can be used lo describe the behavior of electrons under various potential functions. The understanding of electron behavior is crucial in understanding semiconductor physics. The wave-particle duality principle is an important element in quantum mechanics. Particles can have wave-like behavior and waves can have particle-like behavior. Schrodingers wave equation forms the basis for describing and predicting the behavior of electrons- Max Born postulated that 1(л)Р is a probability density function. A result of applying Schrodingers wave equation to a bound particle is that the energy of the bound particle is quantized, A result of applying Schrodingers wave equation to an electron incident on a potential barrier is that there is a finite probability of turmeUng. The basic structure of the periodic table is predicted by applying Schrodingers wave equation to the one-electron atom. GLOSSARY OF IMPORTANT TERMS de Broglie wavelength The wavelength of a particle given as the ratio of Plancks constant to momentum. Heisenberg uncertainty principle The principle that states that we cannot describe with absolute accuracy the relationship between sets of conjugate variables that describe the behavior of particles, such as momentum and position. FauH exclusion principle The principle that states that no two electrons can occupy the same quantum state, photon The particle-like packet of electromagnetic energy, quanta The particle-like packet of themial radiation. quantized energies The allowed discrete energy levels that bound particles may occupy, quantum numbers a set of numbers that describes the quantum state of a particle, such as an electron in an atom. quantum state A particular state of an electron that may be described, for example, by a set of quantum numbers, tunneling The quantum mechanical phenomenon by which a particle may penetrate through a thin potential barrier. wave-particle duality The characteristic by which electromagnetic waves sometimes exhibit particle-Икс behavior and particles sometimes exhibit wave-like behavior CHECKPOINT After studying this chapter, the reader should have the ability to: Discuss the principle of energy quanta, the wave-particle duality principle, and the uncertainty principle. Apply Schrodingers wave equation and boundary conditions to problems with various potential functions. Determine quantized energy levels of bound particles. Determine the approximate tunneling probability of a particle incident on a potential barrier Problems 51  REVIEW QUESTIONS 1. State the wave-particle duality principle and state the relationship between momentum and wavelength. 2. What is the physical meaning of Schrodingers wave function? 3. What is rneant by a probability density function? 4. List the boundary conditions for solutions to Schrodinger*s wave equation. 5. What is meant by quantized energy levels? 6. Describe the concept of tunneling. 7. List the quantum numbers of the one-electron atom and discuss how they were developed. PROBLEMS 2Л The classical wave equation for a two-wire transmission line is given by dVix, t)/dx LC aV(x, /)/a?. One possible solution is given by V{x, t) = (sin Kx) * (sino)/) where К = пл/а and со = К Тс, Sketch, on the same graph, the function V{x, t) as a function of x for 0 < x <a and n 1 when (/) wt = 0, (n) Ш - njl, (Hi) cot ~7T, (/v) cjt = 37Г/2, and (r) cot = 27г. 2.2 The function V{x. t) = cos (2лx -. - o)t) is also a solution to the classical wave equation. Sketch on the same graph the function V(a, r) as a function of д: for -JlJI 0<x <3X when: (0o)t = 0, {ti)cot = 0.25я, (iii)cot = 0.5я, (iv)cot = 0.75я, and = {v)o)t = 7Г. 2.3 Repeat Problem 2.2 for the function V(x, t) = cos (2nx/X + cot). 2.4 Determine the phase velocities of the traveling waves described in Problems 2.2 and 2.3. Section 2Л Principles of Quantum Mechanics 2.5 The work function of a material refers to the minimum energy required to remove an electron from the material. Assume that the work function of gold is 4.90 eV and that of cesium is 1.90 cV. Calculate the maximum wavelength of light for the photoelectric emission of electrons for gold and cesium. 2.6 Calculate the de Broglie wavelength, Xh/p, for: {a) An electron with kinetic energy of (0 1.0 eV, and {ii) 100 eV. (b) A proton with kinetic energy of 1.0 eV. (c) A singly ionized tungsten atom with kinetic energy of LO eV. (d) A 2000-kg truck traveling at 20 m/s. 2.7 According to classical physics, the average energy of an electron in an electron gas at thermal equilibrium is 3kT/2, Determine, for T = 300 K, the average eleclron energy (in eV), average electron momentum, and the de Broglie wavelength. *2.8 An electron and a photon have the same energy. At what value of energy (in eV) will the wavelength of the photon be 10 times that of the electron? 2.9 (a) An electron is moving with a velocity of 2x10* cm/s. Determine the electron energy (in eV), momentum, and de Broglie wavelength (in A), [h) The dc Broglie wave-length of an electron is 125 A. Determine the electron energy (in eV), momentum, and velocity. 2Л0 It is desired to produce x-ray radiation with a wavelength of I A. {a) Through what potential voltage difference must the electron be accelerated in vacuum so that it can. upon coUiding with a target, generate such a photon? (Assume that alt of the electrons energy is transferred to the photon.) (h) What is the dc Broglie wavelength of the electron in part (a) just before it hits the target? 2.11 When the uncertainty principle is considered, it is not possible to locate a photon in space more precisely than about one wavelength. Consider a photon with wavelength X= 1 дт. What is the uncertainty in the photons {a) momentum and (/?) energy? 2.12 The uncertainty in position is 12 A for a particle of mass 5 x 10~- kg. Determine the minimum uncertainty in (a) the momentum of the particle and (h) the kinetic energy of the particle. 2.13 Repeat Problem 2.12 for a pardcle of mass 5 x 10 - kg. 2.14 An automobile has a mass of 1500 kg. What is the uncertainty in the velocity (in miles per hour) when its center of mass is located with an uncertainty no greater than 1 cm? 2.15 {a) The uncertainty in the position of an electron is no greater than 1 A. Determine the minimum uncertainty in its momentum, (b) The electrons energy is measured with an uncertainty no greater than 1 eV. Determine the minimum uncertainty in the time over which the measurement is made. Section 2.2 Schrodingers Wave Equation 2.16 Assume that \{x,t) and fjix t) are solutions of the one-dimensional time-dependent Schrodingers wave equation, (a) Show that + is a solution, (b) Is Ф] - a solution of the Schrodingers equation in general? Why or why not? 2.17 Consider the wave function Ф(л, t) - Л (sin лх)е~ for -1 < < +1. Determine A so that / j IФ(x, t)\- dx 1, 2.18 Consider the wave function Ф(х /) = A(sin пях)е~- for 0 < < 1. Determine A so that j; \>(x,t)\-dx = 1. 2Л9 The solution lo Schrodingers wave equation for a particular situation is given by i/(jc) = V2/ o * Determine the probability of finding the particle between the following Hmits: (a) 0 <x < о/4, ф) /4 Sx < йо/2, and (с) О <x < a. Section 2.3 Applications of Schrodingers Wave Equation 2.20 An electron in free space is described by a plane wave given by Ф(л, /) j(tt-tf) where k i.5 x 10 m~ and a> = 1.5 x 10- rad/s. (a) Determine the phase velocity of the plane wave, (h) Calculate the wavelength, momentum, and kinetic energy (in eV) of the electron. 2.21 An electron is traveling in the negative x direction with a kinetic energy of 0.015 eV. Write the equation of a plane wave that describes this particle. 2.22 An electron is bound in a one-dimensional infinite potential well with a width of 100 A. Determine the electron energy levels for = 1. 2, 3. 2.23 A one-dimensional infinite potential well with a width of 12 A contains an electron. (a) Calculate the first two energy levels that the electron may occupy, (b) If an electron drops from the second energy level to the first, what is the wavejength of a photon that might be emitted? 2.24 Consider a particle with mass of 10 mg in an infinite potential well 1.0 cm wide, (a) If the energy of the particle is 10 mJ, calculate the value of n for that state, {b) What is V(x) к V(x) к Figure 2.11 I Potential function for Problem 2.26. Incident particles л = 0 Figure 2.12 I Potential function for Problem 2.30. the kinetic energy of the ( + 1) state? (c) Would quantum effects be observable for this particle? 2.25 Calculate the lowest energy level for a neutron in a nucleus, by treating it as if it were in an infinite potential well of width equal to 10 m. Compare this with the lowest energy level for an electron in the same infinite potenfial well. 2.26 Consider the particle in the infinite potential well as shown in Figure 2 Л L Derive and sketch the wave functions corresponding to the four lowest energy levels. (Do not normahze the wave functions.) *2.27 Consider a three-dimensional infinite potenfial well. The potential function is given by V{x) = 0 for0 < Л' < a, 0 < у < Of, 0 < г < and V(x) = oo elsewhere. Start with Schrodingers wave equation, use the separation of variables technique, and show that the energy is quantized and is given by 5 + +) 2.29 where = 1,2, 3,..., л^, = 1, 2, 3,..., л, 1, 2, 3..... *2.28 Consider a free electron bound within a two-dimensional infinite potential well defined by V = 0 for 0 < jc < 25 A, 0 < у < 50 A, and = oo elsewhere. Determine the expression for the allowed electron energies. Describe any similarities and any differences to the results of the one-dimensional infinite potential well. Consider a proton in a one-dimensional infinite potential well shown in Figure 2.5. (a) Derive the expression for the allowed energy states of the proton, {b) Calculate the energy difference (in units of eV) between the lowest possible energy and the next higher energy state for (/) a = 4 A, and (/0 й = 0.5 cm. For the step potential function shown in Figure 2.12, assume that E > and that particles are incident from the -\-x direction traveling in the -x direction, {a) Write the wave solutions for each region, {b) Derive expressions for the transmission and reflection coefficients. Consider the penetration of a step potential function of height 2.4 eV by an electron whose energy is 2.1 eV. Determine the relative probability of finding the electron at 2.31 2.32 2.33 the distance (a) 12 A beyond the barrier, and (h) 48 A beyond the barrier, compared to the probability of finding the incident particle at the barrier edge. Evaluate the transmission coefficient for an electron of energy 2,2 eV impinging on a potential barrier of height 6.0 eV and thickness 10~ m. Repeal the calculadon for a barrier thickness of 10 * m. Assume that Equation (2.62) is valid. (a) Estimate the tunneling probability of a particle with an effective mass of 0.067 mo (an electron in gallium arsenide), where mg is the mass of an electron, tunneling through a rectangular potenfial barrier of height Vy =0.8 eV and width 15 A. The particle kinetic energy is 0.20 eV. {h) Repeat part {a) if the effecfive mass of the particle is 1 Шшо (an electron in silicon). A proton attempts to penetrate a rectangular potential barrier of height 10 MeV and thickness 10 m. The particle has a total energy of 3 MeV. Calculate the probability thai the particle will penetrate the potential barrier Assume that Equation (2.62) is valid. *2.35 An electron with energy E is incident on a rectangular potential barrier as shown in Figure 2.8. The potential barrier is of width a and height Vq > £. (й) Write the form of the wave function in each of the three regions, (b) For this geometry, determine what coefficient in the wave function solutions is zero, (c) Derive the expression for the transmission coefficient for the electron (tunneling probability), (d) Sketch the wave function for the electron in each region. *2,36 A potential function is shown in Figure 2.13 with incident particles coming from - oo with a total energy E > V2. The constants к are defined as 2.34 k, = 2m E (£-V,) (£ - v2) Assume a special case for which /:2 =2ля, n=l,2,3, Derive the expression, in terms of the constants, кхЛг. and ky, for the transmission coefficient. The transmission coefficient is defined as the ratio of the flux of particles in region III to the incident flux in region 1. *2.37 Consider the one-dimensional potential function shown in Figure 2.14. Assume the tola! energy of an electron is £ < Vo. {a) Write the wave solutions that apply in each V{x) = ОС Incident particles £ > jr = 0 v = a .v = 0 x = a Figure 2ЛЗI Potential function for Problem 2.36. Figure 2.141 Potential function for Problem 2.37. СНА ВС Т Е R  Introduction to the Quantum Theory of Solids PREVIEW In the last chapter, we applied quantum mechanics and Schrodingers wave equation to determine the behavior of electrons in the presence of various potential functions. We found that one impt>rtant characteristic of an electron bound to an atom or bound within a finite space is that the electron can take on only discrete values of energy; that is, the energies are quantized. We also discussed the Pauli exclusion principle, which stated that only one electron is allowed to occupy any given quantum state. In this chapter, we will generalize these concepts to the electron in a crystal latrice. One of our goals is to determine the electrical properties of a semiconductor material, which we will then use to develop the current-voltage characteristics of semiconductor devices. Toward this end, we have two tasks in this chapter: to determine the properties of electrons in a crystal lattice, and to determine the statistical characteristics of the very large number of electrons in a crystal. To start, we will expand the concept of discrete allowed electron energies that occur in a single atom to a band of allowed ehctron energies in a single-crystal solid. First we will qualitatively discuss the feasibility of the allowed energy bands in a crystal and then we will develops more rigorous mathematical derivation of this theory using Schrodingers wave equation. This energy band theory is a basic principle of semiconductor material physics and can also be izsed to explain differences in electrical characterisrics between metals, insulators, and semiconductors. Since current in a solid is due to the net flow of charge, it is important to determine the response of an electron in the crystal to an applied external force, such as an electric field. The movement of an electron in a lattice is different than that of an electron in free space. We will develop a concept allowing us to relate the quantum me-chanical behavior of electrons in a crystal to classical Newtonian mechanics. This 3.1 Allowed and Forbidden Energy Bands analysis leads to a parameter called the electron effective mass. As part of this development, we will find that we can define a new particle in a semiconductor called a hole. The motion of both electrons and holes gives rise to currents in a semiconductor. Because the number of electrons in a semiconductor is very large, it is impossible 10 follow the motion of each individual particle. We will develop the statistical behavior of electrons in a crystal, noting that the Pauli exclusion principle is an important factor in determining the statistical law the electrons must follow. The resulting probability function will determine the distribution of electrons among the available energy states. The energy band theory and the probability function will be used extensively in the next chapter, when we develop the theory of the semiconductor in equilibrium. 3.11 ALLOWED AND FORBIDDEN ENERGY BANDS In the last chapter, we treated the one-electron, or hydrogen, atom. That analysis showed that the energy of the bound electron is quantized: Only discrete values of electron energy are allowed. The radial probability density for the electron was also determined. This function gives the probability of finding the electron at a particular distance from the nucleus and shows that the electron is not localized at a given radius. We can extrapolate these single-atom results to a crystal and qualitatively derive the concepts of allowed and forbidden energy bands. We can then apply quantum mechanics and Schrodingers wave equation to the problem of an electron in a single crystal. We find that the electronic energy states occur in bands of allowed slates that are separated by forbidden energy bands. ЗЛЛ Formation of Energy Bands Figure 3.1a shows the radial probability density function for the lowest electron energy state of the single, noninteracting hydrogen atom, and Figure 3.1b shows the same probability curves for two atoms that are in close proximity to each other. The wave functions of the two atom electrons overlap, which means that the two electrons   с с о Ш n = 1 n = 1 Figure 3.11 (a) Probability density function of an isolated hydrogen atom, (b) Overlapping probability density functions of two adjacent hydrogen atoms, (c) The splitting of the и = I state. will interact. This interaction or perturbation results in the discrete quantized energy level splitting into two discrete energy levels, schematically shown in Figure ЗЛс. The splitting of the discrete state into two states is consistent with the Pauli exclusion principle. A simple analogy of the splitting of energy levels by interacung particles is the following. Two identical race cars and drivers are far apart on a race track. There is no interaction between the cars, so they both must provide the same power to achieve a given speed. However, if one car puUs up close behind the other car, there is an interaction called draft. The second car will be pulled to an extent by the lead car. The lead car will therefore require more power to achieve the same speed, since it is pulling the second car and the second car will require less power since it is being pulled by the lead car. So there is a splitting of power (energy) of the two interacting race cars. (Keep in mind not to take analogies too literally.) Now, if we somehow start with a regular periodic arrangement of hydrogen-type atoms that are initially very far apart, and begin pushing the atoms together, the initial quantized energy level will split into a band of discrete energy levels. This effect is shown schematically in Figure 3.2, where the parameter ro represents the equilibrium interatomic distance in the crystal. At the equilibrium interatomic distance, there is a band of allowed energies, but within the allowed band, the energies are at discrete levels. The Pauli exclusion principle states that the joining of atoms to form a system (crystal) does not alter the total number of quantum states regardless of size. However, since no two electrons can have the same quantum number the discrete energy must split into a band of energies in order that each electron can occupy a distinct quantum state. We have seen previously that, at any energy level, the number of allowed quantum states is relatively small. In order to accommodate all of the electrons in a crystal, then, we must have many energy levels within the allowed band. As an example, suppose that we have a system with 10* one-electron atoms and also suppose that, at the equilibrium interatomic distance, the width of the allowed energy band is 1 eV. For simplicity, we assume that each electron in the system occupies a different energy level and, if the discrete energy states are equidistant, then the energy levels are separated by 10~ eV. This energy difference is extremely small, so that for all practical purposes, we have a quasi-continuous energy distribution through the allowed с с о  0 Interatomic distance Figure 3.2 I The splitting of an energy state into a band of allowed energies. 1 ... 3 4 5 6 7 8 9 ... 55 |
|
© 2026 AutoElektrix.ru
Частичное копирование материалов разрешено при условии активной ссылки |