 |
 |
 |
| Главная Журналы Популярное Audi - почему их так назвали? Как появилась марка Bmw? Откуда появился Lexus? Достижения и устремления Mercedes-Benz Первые модели Chevrolet Электромобиль Nissan Leaf |
Главная » Журналы » Absorbing materialorganic polymer 1 ... 4 5 6 7 8 9 10 ... 55 Objective example зл To calculate the change in kinetic energy of an electron when the velocity changes by a small value. Consider an electron travehng at a velocity of 10 cm/s. Assume the velocity increases by a value of 1 cm/s. The increase in kinetic energy is given by Let V2 = v\ + Av. Then vl = (i-i + Av)- = i;? + 2vi Av + (Ли) But < iJb so we have that AE milvi Av) = mv\ Av Solution Substituting the number into this equation, we obtain Д£ = (9.11 X 10-)(10-)(0.0i) = 9.11 X !0-* J which may be converted to units of electron volts as 9.11 X 10- AE = -- = 5.7 X 10- eV L6 X 10-1 Comment A change in velocity of 1 cm/s compared with 10 cm/s results in a change in energy of 5.7 X 10~ eV, which is orders of magnitude larger than the change in energy of 10~ eV between energy states in the allowed energy band. This example serves to demonstrate that a difference in adjacent energy states of 10~ eV is indeed very small, so that the discrete energies within an allowed band may be treated as a quasi-continuous distribution. Consider again a regular periodic arrangement of atoms, in which each atom now contains more than one electron. Suppose the atom in this imaginary crystal contains electrons up through the n = 3 energy level. If the atoms are initially very far apart, the electrons in adjacent atoms will not interact and will occupy the discrete energy levels. If these atoms are brought closer together, the outermost electrons in the rt = 3 energy shell will begin to interact initially, so that this discrete energy level will spHt into a band of allowed energies. If the atoms continue to move closer together, the electrons in the л = 2 shell may begin to interact and will also split into a band of allowed energies. Finally, if the atoms become sufficiently close together, the innermost electrons in the = 1 level may interact, so that this energy level may also split into a band of allowed energies. The splitting of these discrete energy levels is energy band. The fact that 10 eV is a very small difference between two energy states can be seen from the following example. a о о   /1 = 3 о Interatomic distance Figure 3.3 I Schematic showing the spHtting of three energy states into allowed bands of energies. = 2 8 electrons  n = I 2 electrons  Six allowed levels at same energy Two allowed levels at same energy с ч> с states О electrons  4N states 4yv electrons 6jV states 2N ekctrom 2Л/ states 2Л^ electrons Figure 3.4 f (a) Schematic of an isolated sihcon atom, (b) The sphtting of the 3s and 3p states of silicon into the allowed and forbidden energy bands. (From Shocktey [5J.) qualitatively shown in Figure 3.3. If the equilibrium interatomic distance is ro, then we have bands of allowed energies that the electrons may occupy separated by bands of forbidden energies. This energy-band splitting and the formation of allowed and forbidden bands is the energy-band theory of single-crystal materials. The actual band splitting in a crystal is much more complicated than indicated in Figure 3.3. A schematic representation of an isolated silicon atom is shown in Figure 3.4a. Ten of the fourteen silicon atom electrons occupy deep-lying energy levels close to the nucleus. The four remaining valence electrons are relatively weakly bound and are the electrons involved in chemical reactions. Figure 3.4b shows the band splitting of silicon. We need only consider the ft = 3 level for the valence electrons, since the first tw-o energy shells are completely full and are tightly bound to the nucleus. The 3s state corresponds to n = 3 and / = 0 and contains two quantum states per atom. This state will contain two electrons at Г = 0 K. The 3p state corresponds to n = 3 and / = 1 and contains six quantum states per atom. This state will contain the remaining two electrons in the individual silicon atom. As the interatomic distance decreases, the 3s and 3p states interact and overlap. At the equilibrium interatomic distance, the bands have again split, but now four quantum states per atom are in the lower band and four quantum states per atom are in the upper band. At absolute zero degrees, electrons are in the lowest energy state, so that all states in the lower band (the valence band) will be full and all states in the upper band (the conduction band) will be empty. The bandgap energy Eg between the lop of the valence band and the bottom of the conduction band is the width of the forbidden energy band. We have discussed qualitatively how and why bands of allowed and forbidden energies are formed in a crystal. The formation of these energy bands is directly related to the electrical characteristics of the crystal, as we will see later in our discussion. *3.1.2 The Kronig-Penney Model In the previous section, we discussed qualitatively the spitting of allowed electron energies as atoms are brought together to form a crystal. The concept of allowed and forbidden energy bands can be developed more rigorously by considering quantum mechanics and Schrodingers wave equation. It may be easy for the reader to *get lost in the following derivation, but the result forms the basis for the energy-band theory of semiconductors. The potential function of a single, noninteracting, one-electron atom is shown in Figure 3.5a. Also indicated on the figure are the discrete energy levels allowed for the electron. Figure 3.5b shows the same type of potential function for the case when several atoms are in close proximity arranged in a one-dimensional array. The potential functions of adjacent atoms overlap, and the net potential function for this case is shown in Figure 3.5c. It is this potential function we would need to use in Schrodinger*s wave equation to model a one-dimensional single-crystal material. The solution to Schrodingers wave equation, for this one-dimensional single-crystal lattice, is made more tractable by considering a simpler potential function. Figure 3.6 is the one-dimensional Kronig-Penney model of the periodic potential function, which is used to represent a one-dimensional single-crystal lattice. We need to solve Schrodingers wave equation in each region. As with previous quantum mechanical problems, the more interesting solution occurs for the case when £ < Vq, which corresponds to a particle being bound within the crystal. The electrons are contained in the potential wells, but we have the possibility of tunneling between wells. The Kronig-Penney model is an idealized periodic potential representing a one-dimensional single crystal, but the results will illustrate many of the important features of the quantum behavior of electrons in a periodic lattice. To obtain the solution to Schrodingers wave equation, we make use of a mathematical theorem by В loch. The theorem states that all one-electron wave functions. indicates sections that can be skipped without loss of continuity. V = 0  v(r)a i  Atom Atom Atom Atom Atom Atom Atom Atom Figure 3.5 I (a) Potential function of a single isolated atom, (b) Overlapping potential functions of adjacent atoms, (c) Net potential function of a one-dimensional single crystal.  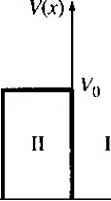   (rt + b) -h a (й + b) Figure 3.6 I The one-dimensional periodic potential function of the Kronig-Penney model. for problems involving periodically varying potential energy functions, must be of the form lAU) u(x)eJ (3.1) The parameter к is called a constant of motion and will be considered in more detail as we develop the theory. The function u(x) is a periodic function with period We stated in Chapter 2 that the total solution to the wave equation is the product of the time-independent solution and the time-dependent solution, or which may be written as Cvл) - w(.r)e-/ />> (3.3) This traveling-wave solution represents the motion of an electron in a single-crystal material. The amplitude of the traveling wave is a periodic function and the parameter jt is also referred to as a wave number. We can now begin to determine a relation between the parameter k, the total energy £, and the potential vq. If we consider region 1 in Figure 3.6 (0 < л < о) in which V{x) - 0, take the second derivarive of Equation (3.1), and substitute this result into the time-independent Schrodingers wave equation given by Equation (2.13), we obtain the relation (f-uxix) , du\(x) -y + V-~ - i}c - a)u,{x) - 0 (3.4) dx dx The function (.r) is the amplitude of the wave function in region 1 and the parameter a is defined as о 2m E a- - (3.5) Consider now a specific region II, -b < x < 0, in which V(x) Vq, and apply Schrodinger4 wave equafion. We obtain the relation d-U2ix) du2(x) / . . 2mVo\ , , + ~dx--\ ~~lf~ yi) = () where U2{x) is the amplitude of the wave function in region П. We may define 2m 2mV{) - {E vo) = = (i (3.7) so that Equation (3.6) may be written as - + 2jk~ ik рЪи.Лх) - 0 (3,8) dx dx Note that from Equation (3.7), if £ > Vo the parameter is real, whereas if £ < Vo, then is imaginary. .vO X (3.13) dx we obtain (a - k)A - (a + k)B -ifi- k)C + ( + k)D 0 (3.14) We have considered region 1 as 0 < < a and region 11 as -7 < x < 0. The periodicity and the conrinuity condition mean that the function Н]. as x й, is equal to the function ui, as Jt -/?. This condition may be written as ui(a) = U2(-b) (3.15) Applying the solutions for ui(x) and U2(x) to the boundary condition in Equation (3.15) yields The last boundarv condition is (3.17) which gives {a - k)AeJ - - (a + k)Be- - (j9 - k)Ce- -\-ф-\г k)DeJ 0 (3.18) We now have four homogeneous equation.s. Equations (3.12), (3.14), (3.16), and (3.18), with four unknowns as a result of applying the four boundary conditions. In a set of simultaneous, linear, homogeneous equations, there is a nontrivial solution if. The solution to Equation (3.4), for region 1, is of the form и, (л) = Ле- + Ве- - for (О < х < а) (3.9) and the solution to Equation (3.8), for region 11, is of the form ii2{x) = Ce- + 0--+ for (-b < X < 0) (3.10) Since the potential function V{x) is everywhere finite, both the wave function \p[x) and its first derivative д'ф(х)/дх must be continuous. This continuity condition implies that the wave amplitude function u(x) and its first derivative du(x)/Bx must also be continuous. If we consider the boundary at Jt = 0 and apply the continuity condition to the wave amplitude, we have w,(0) = ш(0) (3.11) Substituting Equations (3,9) and (3.10) into Equation (3,11), we obtain Л + O (3.12) Now applying the condition that and only if, the determinant of the coefficients is zero. In our case, the coefficients in quesdon are the coefficients of the parameters A, B, C, and D. The evaluafion of this determinant is extremely laborious and will not be considered in detail. The result is -----(sin Of a) (sin jSb) + (cos oa) (cos 3?) = cosk{a +/?) (3.19) Equafion (ЗЛ 9) relates the parameter к to the total energy В (through the parameter a) and the potenfial function Vq (through the parameter p). As we mentioned, the more interesting solutions occur for £ < Vy, which applies to the eiectron bound within the crystal. From Equation (3.7), the parameter is then an imaginary quantity. We may define Pjy (3-20) where у is a real quantity. Equation (3.19) can be written in terms of у as У - О (sinart)(sinhy) -h (cos aa) (cosh у b) = cos к (a -i-b) (3.21) 2a у Equation (3.21) does not lend itself to an analytical solution, but must be solved using numerical or graphical techniques to obtain the relation between £, and Vy The solution of Schrodingers wave equation for a single bound particle resulted in discrete allowed energies. The solution of Equation (3.21) will result in a band of allowed energies. To obtain an equation that is more susceptible to a graphical solution and thus will illustrate the nature of the results, let the potential barrier width b 0 and the barrier height Vq oo, but such that the product Vq remains finite. Equation (3.21) then reduces to т¥фа\ siiiaa --T---\-сонаа - cos/c (3.22) / aa We may define a parameter P as m Vfiba P - (3.23) Then, finally, we have the relation .sin aa P---- + cosaa = cos ka (3.24) Equation (3.24) again gives the relation between the parameter k, total energy E {through the parameter a), and the potential barrier bVo. We may note that Equation (324) is not a solution of Schrodingers wave equation but gives the conditions for which Schrodingers wave equation will have a solution. If we assume the crystal is infinitely large, then к in Equation (3.24) can assume a continuum of values and must be real. ЗЛ J The ft-Space Diagram To begin to understand the nature of the solution, initially consider the special case for which Vq - 0, In this case P - 0, which corresponds to a free particle since there are no potential barriers. From Equation (3.24), we have that cosaa = cos ka a = к (3.25) (3-26) Since the potential is equal to zero, the total energy E is equal to the kinetic energy, so that, from Equation (3.5), Equation (3.26) may be written as 2mE 2m{mv-) p (3.27) where p is the particle momentum. The constant of the motion parameter к is related to the particle momentum for the free electron. The parameter is also referred to as a wave number. We can also relate the energy and momentum as (3.28) Figure 3.7 shows the parabolic relation of Equation (3.28) between the energy £ and momentum p for the free panicle. Since the momentum and wave number are Hn-early related. Figure 3.7 is also the E versus к curve for the free particle. We now want to consider the relation between E and к from Equation (3.24) for the particle in the single-crystal lattice. As the parameter P increases, the particle I becomes more tightly bound to the potential well or atom. We may define the left side of Equation (3.24) to be a function fipta), so that ,smaa P--+ cosQfa (3.29) I I I f /7 = 0 pox к Figure 3.7 1 The parabolic E versus к curve for the free electron. sin aa 
fiaa) ka = Itt ka =0
ka = -IT ka - 7T ka ~ Ътт Figure 3.8 IA plot of (a) the first term in Equation (3,29), (b) the second term in Equafion (3.29), and (c) the entire fiaa) function. The shaded areas show the allowed values of {aa) corresponding to real values of k. Figure 3.8a is a plot of the first term of Equation (3.29) versus aa. Figure 3.8b shows a plot of the cos aa term and Figure 3.8c is the sum of the two terms, or /(aa). Now from Equation (3.24), we also have that f{aa) = cos ka (3.30) For Equation (3.30) to be valid, the allowed values of the /(ofa) function must be bounded between +1 and -1. Figure 3.8c shows the allowed values of f(aa) and the allowed values ofaa in the shaded areas. Also shown on the figure are the values of fl from the right side of Equation (3.30) which correspond to the allowed values Gffiaa). The parameter a is related to the total energy E of the particle through Equafion (3.5), which is a - 2mE/fi, A plot of the energy E of the particle as a function of the wave number к can be generated from Figure 3.8c. Figure 3.9 shows this plot and shows the concept of allowed energy bands for the particle propagating in the  Figure 3,91 The E versus к diagram generated from Figure 3.8. The allowed energy bands and forbidden energy bandgaps are indicated. crystal lattice. Since the energy E has discontinuities, we also have the concept of forbidden energies for the particles in the crystal. EXAMPLE 3.2 Objective To determine the lowest allowed energy bandwidth. Assume that the coefficient я = 10 and that the potenfial width й = 5 A. Solution To find the lowest allowed energy bandwidth, we need to find the difference in aa values as ka changes from 0 to л: (see Figure 3.8c)- For ka = 0, Equation (3.29) becomes sin aa 1 10---h cosaa By trial and error, we find aa = 2.628 rad. We see that for ka л.аа For aa = л, we have 2m E- E, = jr(1.054 x 10-) 2ma- ~ 2(9.11 x 10--)(5 x 10-) = 2.407 X 10-* J = 1.50 eV For aa = 2.628, we find that £i = 1.68 x 10 J = 1.053 eV. The allowed energy bandwidth is then AE = E2 - El 1.50- 1.053 = 0.447 eV 1 ... 4 5 6 7 8 9 10 ... 55 |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
© 2026 AutoElektrix.ru
Частичное копирование материалов разрешено при условии активной ссылки |